Volume 12 (2024)
Volume 11 (2023)
Volume 10 (2022)
Volume 9 (2021)
Volume 8 (2020)
Volume 7 (2019)
Volume 6 (2018)
Volume 5 (2017)
Volume 4 (2016)
Volume 3 (2015)
Volume 2 (2014)
Volume 1 (2013)
Keywords = laccase
Number of Articles: 1
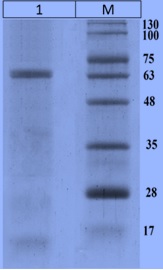
Recombinant CotA-Laccase from Bacillus Licheniformis: Characterization and Overexpression in Bacillus Subtilis WB600
Volume 11, Issue 4 , October 2023, , Pages 196-205